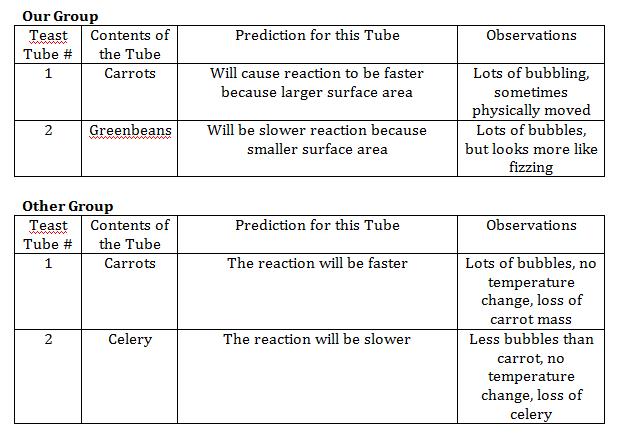
Background
Chemical reactions are an essential part of almost all living things. Living things are constantly moving and changing requiring quick chemical reactions. Enzymes fill the need for something to speed up this job. Enzymes are a type of protein with variations in shapes and sizes each specifically designated for a specific job. Their jobs are to either break down materials (substrate) or changing the function of it. All of them all do the same job of lowering the activation energy required for a chemically reaction allowing for a faster reaction. There are stipulations to this though. The three main environmental ones are temperature, pH level, and salinity, each of which can change the enzymes shape causing it to not be able to do it’s job or doing it differently. There are other factors as fake coenzymes and inhibitors that bind to the active site also changing the enzymes shape or blocking substrate from being processed.
Catalase is a type enzyme found in most living things. It as other enzymes performs a specific job. That specific job is to break down hydrogen peroxide that is produced by aerobic respiration in the cells. Hydrogen peroxide is dangerous to living things so if not removed the cells that produce it will not be able to survive. Basically it breaks down hydrogen peroxide into to smaller compounds, oxygen gas and water.
Equation2H2O2 --> 2H2O + O2
Pre-Lab Data
Catalase is a type enzyme found in most living things. It as other enzymes performs a specific job. That specific job is to break down hydrogen peroxide that is produced by aerobic respiration in the cells. Hydrogen peroxide is dangerous to living things so if not removed the cells that produce it will not be able to survive. Basically it breaks down hydrogen peroxide into to smaller compounds, oxygen gas and water.
Equation2H2O2 --> 2H2O + O2
Pre-Lab Data
Lab-set up
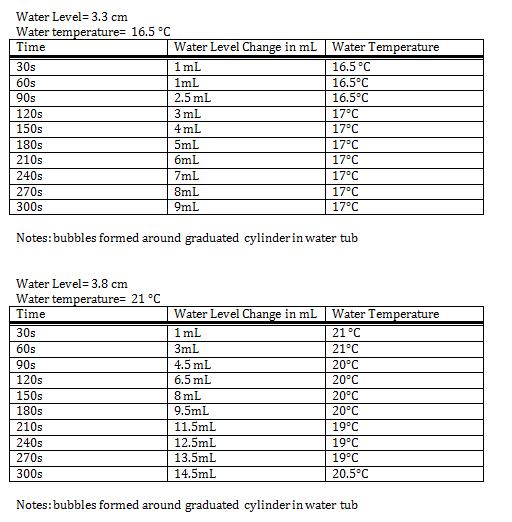
This set up is for a lab to test the influence of environmental factors on an enzymes ability to react. To test, a tub(4) was filled with water till 3.3 cm in depth. Then a graduated cylinder (2) upside down in the water while holding water with no bubbles. The temperature of the water was recorded with a thermostat (1) to measure other uncontrollable variables. Once everything was in place, 10 ml of hydrogen peroxide was measured in a beaker (5) and then poured into the reaction chamber (3). Then 4 mL of the plant catalase was measured and added to the reaction chamber. Once both solutions were added, the cap of the reaction chamber was screwed on and placed in the tub of water pointing with the nozzle end under the graduated cylinder (6) to capture the oxygen. The oxygen released is a result of the hydrogen peroxide being converted into water and oxygen gas. The oxygen gas will travel up becoming trapped in the upside down graduated cylinder(7). During the entire reaction process temperature and water displacement in the cylinder were recorded in 30 second intervals every 5 minutes. To test the catalase with a variable 150 mL of hot water was added into the tub. This in turn raised the initial temperature to 21˚C and water level to 3.8 cm.
Data
Initial Rate of Enzyme
Determining a difference between similar enzyme reactions can be difficult, but analyzing an initial rate of a reaction can paint a clearer picture. The initial rate of an enzyme reaction is the reaction speed an enzyme breaks down a substrate in the first several minutes. For this lab, the first 3 minutes will be used to determine the initial rate.
Equation: (M2-M1)/(T2-T1)
Test without hot water
(5-0)/(3-0) = 1.67 mL per minute
Test with hot water
(9.5-0)/(3-0) = 3.167 mL per minute
Equation: (M2-M1)/(T2-T1)
Test without hot water
(5-0)/(3-0) = 1.67 mL per minute
Test with hot water
(9.5-0)/(3-0) = 3.167 mL per minute
Conclusion
The Results from the test without hot water (part A) compared to the results the test with hot water (part B) are quite strong. The addition of the hotter water made a large increase in oxygen gas formation in the graduated cylinder. Even more interestingly towards the end of the test when the water began to cool down a few degrees so did the reaction rate of the hydrogen peroxide with the catalase. This shows that the external variables, such as water temperature play a large role in the reaction rate.
Possible Sources of Error
For the lab, the findings for the conclusion had a risk for errors. One of the possible errors is the water level being different by adding additional water into the tub. This could have lead to a more complete heat dispersion of the water on the reaction chamber giving a different result. Another possible error is that a student had to hold the reaction chamber to ensure the oxygen gas bubbles were going into the graduated cylinder to be recorded. The hand of the student could have released excess heat during the initial test and absorb heat during the second test.